
Do you know about Parkinson’s disease?
Parkinson’s disease is a progressive nervous system disorder that affects movement. Symptoms start gradually, starting with a barely noticeable tremor in just one hand. Now, till simple tremor, it’s fine, almost everyone experiences that somehow or other. However, when the disorder starts causing stiffness or slowing of movement, it’s a sign of seriousness.
Talking about the disorder, the recent studies related to it have surely proved the known saying “The answers lie within you.”
A molecular picture of the problem
The death of the neurons specialized in the synthesis of dopamine greatly affects the cognitive capacities of a person suffering from Parkinson’s disease. Now, this loss of the specialized neurons results in alpha-synuclein aggregation. Alpha-Synuclein (α-S) is a 140 amino acid long protein, which aggregates into amyloid fibrils in a subset of neuronal and glial cells, which lie behind the onset of a group of progressive and, ultimately, fatal neurodegenerative disorders, including Parkinson’s disease (PD) – of which we are talking about here.
The initial aggregates of the alpha-synuclein are known as oligomers – responsible for spreading the disease in the brain. Previous studies have identified two types of oligomers:
- non-toxic type A
- the most harmful type B
along with even larger clumps called fibrils.
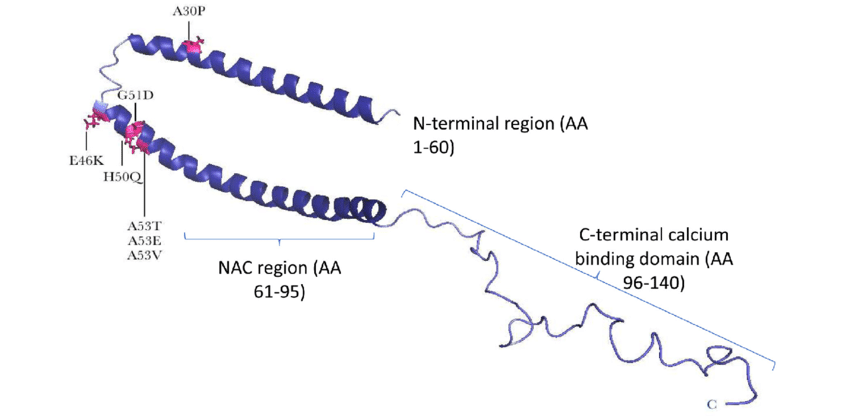
(Source: Whittaker et al., F1000 Research)
Coming to this, a solution clicks!
Why do we not neutralise the oligomers and solve the problem?
It is difficult to do so because of their diverse, dynamic, and transient nature. Yet, several specialized techniques and lab-grown neural cells have proven to be effective in binding with the toxic forms of alpha-synuclein, hence, preventing the conversion of type B oligomer to fibrils somewhat.
So what’s the solution?
Maybe not the oligomers, but an endogenous helical antimicrobial peptide, which is naturally attached to the alpha-synuclein, has our answer. This is called LL-37, also known as, cathelicidin.
Fun fact:
LL-37, despite being a small attachment, has strength superior to any known peptides.
Continuing with the story…
LL-37 has a hydrophobic side and a positively charged side. It interacts with the alpha-synuclein oligomers and inhibits aggregation it goes through, yes! It does so at a very low rate and concentration. However, more importantly, it prevents the damage of neuronal cells. And, that is exactly our key, right?
In short, LL-37 binds with the type B oligomers and fibrils with strong interactions, a comparatively much weaker binding with the type A oligomers, and, no binding at all with the free alpha-synuclein.
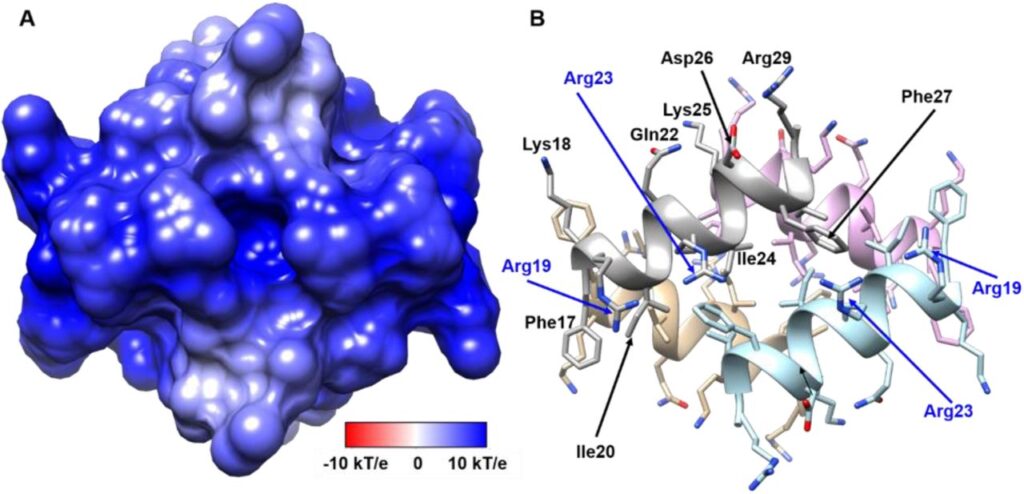
(Source: Engelberg et al., Nat. Comm.)
Encouraged by this new discovery, researchers are planning to study more in relation to its possible therapeutic applications. Until now, scientists could not discover such a molecule that would selectively differentiate between functional and toxic alpha-synucleins. In addition, this molecule can also help in the diagnosis of several other conditions related to alpha-synuclein clumps.
Fun Fact:
Reading about this research, I got another interesting chunk. The main aim of their studies was in relation to alpha-synuclein only. It was only during their thorough study of its structure, that they discovered the magic in-built molecule: LL-37. Why are all successful searches done unknowingly?
Also…
Along with our anti-aggregation molecule, LL-37, which is found in humans naturally, scientists have also found a polypeptide present in bacteria with almost a similar function. And we have been successful in making an artificial molecule with similar functions. Amazing, right?
What do you think, which one will prove to be the most efficient – the human type or the bacterial type? Well, put on your scientific hats and try to find the best answer for humanity.
By,
Shruti Mandal (BS-MS Student, Department of Biological Sciences, IISER Kolkata)
References:
- https://www.mayoclinic.org/diseases-conditions/parkinsons-disease/symptoms-causes/syc-20376055
- https://www.nature.com/articles/s41467-021-24039-2
- https://www.sciencedirect.com/science/article/pii/S0006295215001070
About the author
Shruti Mandal, a sophomore of IISER Kolkata is ongoing her BS-MS course, in the department of biological sciences. She likes to introduce herself as a – ‘Jack of all trades, master of none ‘. She is a regular writer at The Qrius Rhino and you can find all her blogs here.
1 thought on “Naturally found LL-37 : A Savior”