
Cancer is a major global concern and it is generally defined as a type of disease in which there is abnormal cell growth and proliferation, which has the potential to invade or spread to other parts of the body. It can happen to any type of cell in the body and if not diagnosed and treated early, can be fatal. It is the second leading cause of death globally, accounting for an estimated 9.6 million deaths.
Learn more about the different stages and clinical nomenclature of cancer.
Chemotherapeutic drugs cannot distinguish between killing cancer cells and killing microbes. The gut microbes help us to assimilate the food and keep us healthy. Killing good microbes is harmful, especially for children. The evidence shows that disruption of the gut microbiome in early life can lead to potential health conditions later in life. The increasing number of long-term survivors highlights the need to elucidate the underlying mechanisms in chemotherapy-induced late effects.
Learn more about cancer chemotherapy and its side effects.
Recently, animal models and clinical studies have uncovered the significant association between changes in the intestinal microbiota and treatment-related comorbidities. Therefore, the emerging role of the gut microbiome in late effects among cancer survivors is gaining more attention and is very prospective. Longitudinal clinical studies in chemotherapy-treated patients represent a significant challenge.
If you want to know why larger animals do not have a proportionally higher risk of having cancer read the blog on Peto’s Paradox. Also, in case you want to know how smoking/tobacco causes cancer, check out the well-illustrated infographics here.
Gut microbes – the game changer
The gut microbiota has been found to actively interact with the host, and we are just at the beginning of deciphering the exact signals of communication. The enormous microbial diversity among cancer patients suggests that the most efficient therapies may be directed at the unique microbiota composition rather than individual bacterial strains[1].
Chemotherapeutic drugs induce gastrointestinal side effects that may be manipulated by the naturally occurring microbiome. They have the potential to prevent the risk of infections and additional health complications.
Naturally, occurring Gut bacteria can remove the chemo toxins in the body. The good bacteria in the human gut can be great companions. Recently in a Northwestern University study published on May 26, 2021, scientists found that specific types of gut bacteria can safeguard the good bacteria from chemotherapy by alleviating harmful, drug-induced changes to the gut microbiome. Biotransforming of chemotherapeutics can promote resilience in simplified intestinal microbial communities. By metabolizing chemotherapy drugs, the guarding bacteria could mettle short-and long-term side effects of chemotherapy.
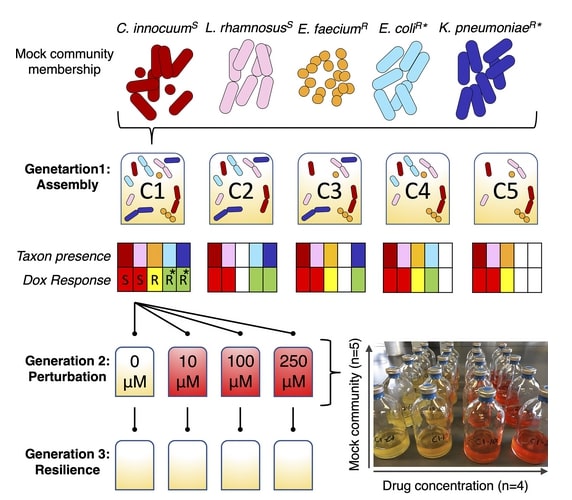
They developed an in-vitro model to characterize fundamental changes in bacterial community dynamics under chemotherapeutic stress and studied the role of the bacterial interactions on drug detoxification to promote microbiota resilience. Raoultella planticola, a naturally occurring gut bacteria present in low quantities can break down chemotherapy drug doxorubicin. To take this further, they wanted to test if Raoultella planticola’s breakdown effect could save the entire microbiome. The scientists developed simplified microbial communities that contain myriad types of bacteria commonly found in the human gut. And then tested the hypothesis that bacteria-mediated detoxification of doxorubicin (most common chemotherapeutic).
The mock communities were made up of five specific members as predicted by genomic analysis. They were sensitive to the drug or resistant via biotransformation and/or efflux were grown invitro over three generations. And characterize the community assembly, response to perturbation (doxorubicin exposure), and resilience.
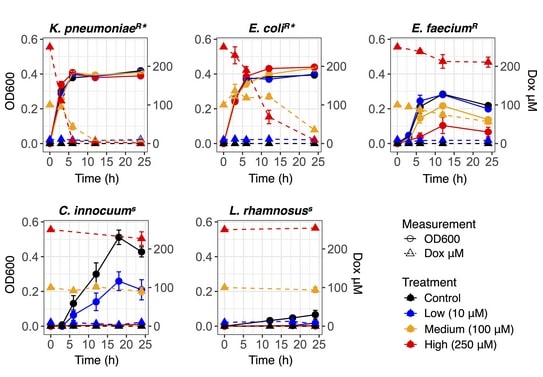
These “mock gut communities” had bacterial strains (Escherichia coli and Klebsiella pneumoniae) that are great at breaking down doxorubicin. The strains (Clostridium innocuum and Lactobacillus rhamnosus) are sensitive to doxorubicin and, one strain (Enterococcus faecium) is immune to doxorubicin but can’t break it down.
They revealed these “mock gut communities” to doxorubicin and saw an increased survival rate among the sensitive strains. They concluded that certain bacteria made the drugs less toxic to the rest of the gut by the degradation of doxorubicin. Spectrophotometric assays were used to monitor the bacterial growth and drug concentrations. 16S rRNA gene sequencing was used to evaluate the relative strain abundances.
The flaring growth of drug-sensitive strains in monoculture by lowering the concentration of doxorubicin in culture media. This predicted the resistance of bacteria involving biotransformation. In mixed communities, the resilience of drug-sensitive members was largely governed by the presence and efficacy of transformers along with the concentration of drug exposure.
The bacteria resistant to doxorubicin also improved these protective effects when the transformers were resistant to doxorubicin via efflux[2]. The simplified community unravelled ecological relationships among a dynamic consortium and highlighted drug detoxification by a keystone species.
This research highlights a promising new pathway for potentially protecting cancer patients. The scientists cautioned that there is still a far way in translating these new findings into treatments. The research could significantly help pediatric patients, but there is still a long way to make this a reality.
In the meantime, knowing how naturally occurring gut bacteria can temper chemotherapy toxins that might lead to better ways to treat chemotherapy side effects for everyone, and that’s the cutting edge!
By Sri Sindhu Bhatta (1st-year BS-MS student, IISER Kolkata)
Read also:
- Peto’s Paradox – An Answer to Cancer Therapy?
- World ‘NO’ Tobacco Day – why tobacco causes cancer?
- Stages of Cancer
- Cancer Chemotherapy
References
- Cancer – World Health Organisation (WHO) (accessed on 02 June 2021)
- Ciernikova, Sona; Mego, Michal; Chovanec, Michal. 2021. “Exploring the Potential Role of the Gut Microbiome in Chemotherapy-Induced Neurocognitive Disorders and Cardiovascular Toxicity” Cancers 13, no. 4: 782. https://doi.org/10.3390/cancers13040782
- Blaustein RA, Seed PC, Hartmann EM. Biotransformation of doxorubicin promotes resilience in simplified intestinal microbial communities. mSphere. 2021. doi: 10.1128/mSphere.00068-21
About the author:

Sri Sindhu Bhatta is a freshman at IISER Kolkata. She is a science enthusiast keen on bioengineering. Aspires to pursue research in the interface between medicine and AI. Simply put she’s always seen to dabble around many things. She loves singing with her ukulele and occasionally tries new projects with Arduino. A talented Table tennis player, an amateur artist, and a bibliophile. She is futuristic and believes that there is more to know.
This is her first article at Qrius Rhino.
1 thought on “Naturally Occurring Gut Bacteria Can Clean Up Chemotherapy Toxins in the Body”